- +62 813 1417 0122
- info@mizuiosmosa.co.id
- Jl. SMP 135 No. 37, Pondok Bambu, Duren Sawit, Jakarta Timur 13430

Welcome to our “field lecture” today. If you are reading this article, chances are you are in a remote area like Kalimantan, Sumatra, or Riau, staring at a water source that is reddish-brown like thick tea. As a Senior Water Chemist at PT Mizui Osmosa Teknovasi, I often get questions from engineers in the field: “Sir, why is this water so hard to clear up even after adding a basket of alum?” The answer is not as simple as “not enough chemicals.” Peat water is a unique chemical phenomenon. In this guide, we will deeply dissect how to purify peat water using a correct scientific approach that remains practical to apply in the field.
Before we jump into the solutions, we must understand the characteristics of the water we are dealing with. Many people fail to treat peat water because they assume it is the same as regular turbid river water (surface water containing mud/silica). In reality, they are chemically very different.
Peat water is reddish-brown not because of mud, but due to its high dissolved organic matter content. This color comes from the decomposition of organic materials (leaves, twigs, tree roots) rotting in swamplands for hundreds of years.
Specifically, there are three main components that are the “culprits” in swamp water treatment methods:
Humic Acid and Fulvic Acid: These are complex organic compounds that give a yellow to dark brown color. They are colloidal (suspended and not easily settled) and highly stable. The acidic nature of these compounds causes the pH of peat water to plummet.
Organic Iron (Fe): In peat water, iron is often bound to organic matter, forming organometallic complex compounds. This makes the iron difficult to oxidize using just normal air.
Low Suspended Particles: Unlike turbid river water in Java which is cloudy due to mud, peat water often appears clear (transparent) but colored. This is deceiving to the eye. In reality, its organic content is extremely high (high Permanganate/KMnO4 value).
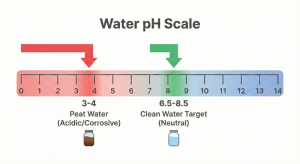
Why should we bother using these red water purification techniques? Why not just use it directly? Operationally and from a health perspective, using raw peat water (pH 3–5) is highly detrimental:
Corrosive: Low pH will rapidly eat away at your iron pipes, pumps, and metal infrastructure.
Skin Health: The highly acidic nature can cause skin irritation, itching, and dermatitis, especially for sweating workers.
Aesthetics and Stains: Iron and organic matter will leave permanent yellow stains on clothes (laundry) and scale on bathroom tiles.
Therefore, referring to clean water quality standards (such as those stated in the Minister of Health Regulation No. 32 of 2017), the pH parameter must be neutralized to 6.5 – 8.5 and the color must be removed.
The first step in how to treat turbid peat water is not to immediately add a purifier, but rather to fix its pH first. This is the most common mistake I encounter in the field.
Many beginner WTP (Water Treatment Plant) operators immediately pour Alum into peat water. The result? The water remains red, the alum simply dissolves, and no flocs are formed.
Chemically, coagulants like Aluminum Sulfate (Alum) work optimally in the pH range of 6.0 to 8.0. If your raw water pH is at 3.5 (highly acidic), the hydrolysis reaction necessary to form floc will not occur. Instead, the alum will remain dissolved and increase the water’s acidity.
Therefore, the very first mandatory chemical for peat water is a pH adjuster. You have two main options:
Quicklime / Hydrated Lime (Ca(OH)2):
Pros: Cheap, easily found in hardware stores.
Cons: Increases water hardness, can cause scaling in pipes if excessive, low solubility (leaves white residue).
Soda Ash (Na2CO3):
Pros: Dissolves perfectly, does not increase hardness, fast reaction.
Cons: More expensive than lime.
The lime dosage for peat water varies depending on the initial acidity level. However, as a rule of thumb for a Jar Test:
Try dissolving 20-50 grams of lime per 1,000 liters of water.
Stir evenly, then check with a pH meter or litmus paper.
Target a pH of at least 6.5 – 7.0 before proceeding to the next stage.

After the water’s pH rises to neutral (around pH 7), only then can we talk about “catching” the red color. This is where the coagulation and flocculation process comes into play.
When choosing a coagulant, there is a classic debate between Alum and PAC.
Alum (Al2(SO4)3): Effective, but requires a narrow pH range. Alum produces more sludge (large but light “flocs”).
PAC (Poly Aluminium Chloride): This is a more versatile modern coagulant. PAC works effectively over a wider pH range and varying temperatures. Its polymer chains can bind organic colloidal particles (peat water color) more strongly and produce denser sludge (lower sludge volume).
For “stubborn” peat water cases, my recommendation is to use PAC. Even though it is more expensive per kg, the required dosage is usually much smaller compared to alum, making it cost-effective.
Mixing chemicals is not just “drop and stir.” Fluid physics comes into play here.
Flash Mixing: When the chemical (PAC) is added, the water must be stirred very rapidly (100-200 rpm) for 1-2 minutes. The goal is to evenly distribute the chemical and instantly neutralize the electrical charge of the dirt particles.
Slow Mixing: After that, reduce the stirring speed to very slow (20-40 rpm) for 15-20 minutes. The purpose is to allow the now-neutralized small particles to collide and merge into large clumps (flocs). If stirred too vigorously at this stage, the flocs will break apart again!
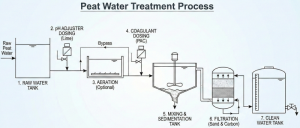
After the sedimentation process where the mud flocs fall to the bottom of the tank, the water on top (supernatant) is usually much clearer and no longer red. However, the process is not finished yet. There is still a potential for bacteria and dissolved iron residue.
Sometimes, the treated water still smells of “iron” or fishy. This is because the iron has not fully oxidized. The functions of alum and PAC are only to bind particles. They do not kill germs.
Add Chlorine (Calcium Hypochlorite) as a disinfectant. Besides killing E. coli bacteria, chlorine also acts as a strong oxidizer that will convert residual dissolved iron into solid particles (rust) that can be filtered.
The final step is physical filtration. Don’t just rely on sponges or palm fiber (ijuk). Use industry-standard filter media arranged in FRP vessels or filter tanks:
Silica Sand: Functions to filter out residual mud flocs that escape the settling tank.
Activated Carbon: This is the key to an organic peat water filter. Activated carbon functions to absorb (adsorb) odor, taste, residual organic color, and excess chlorine residue. Without activated carbon, the water might be clear but still have an unpleasant smell.
The guide above covers basic chemical principles that you can apply with a simple peat water purifier schematic (for example, using a blue barrel or a manual concrete tank). This method is suitable for individual household use or small guard posts.
But, what if you are managing:
A plantation employee mess with 500 residents?
A site plant that needs water for boilers or engine cooling?
Public facilities in an assisted village?
Relying on operators to scatter lime and manually stir every day carries significant risks:
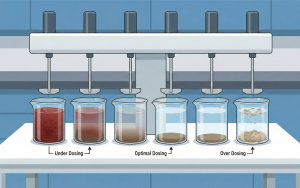
Dosage Inconsistency: Peat water quality fluctuates (during rain vs. drought). Manual dosing is often feeling-based, causing the water to be clear sometimes and still red at others.
Wait Times: The manual settling process is time-consuming (batch system). You cannot get a continuous 24-hour water supply.
Human Error: An operator forgetting to add chlorine can be fatal (diarrhea outbreak in the camp).
This is where PT Mizui Osmosa Teknovasi comes in. We understand that in the field, you need a reliable and “robust” system.
We provide Packaged Water Treatment Plant (WTP) solutions specifically designed for peat water characteristics (Peat Water Series). The advantages of our system include:
Automatic Dosing Pump: A precise chemical dosing pump, ensuring the pH and coagulant are always exact, regardless of who the operator on duty is.
Static Mixer & Flocculator: Hydraulically designed mixing technology, ensuring perfect blending without consuming excessive electricity.
Engineered Filtration: FRP filter tanks with a multi-media bed calculated based on the correct loading rate, not just filled with sand.
Technical Support: Our expert team doesn’t just sell equipment; we provide knowledge transfer and clear SOPs.
If you are tired of employee complaints about itchy bath water or yellowing uniforms, it’s time to switch to a measurable system.
Purifying peat water is the art of balancing chemistry. Start by conducting a small-scale Jar Test using the guide above. However, if you need a more in-depth laboratory water analysis or an industrial-scale WTP system quotation, don’t hesitate to discuss it with us. Contact PT Mizui Osmosa Teknovasi for further consultation.
References:
Butuh bantuan? Tim kami siap membantu.